The criteria for authorship in medical research are vague, often leaving researchers unsure of who belongs in the byline. While there are guidelines for authorship and acknowledgment, in practice, matters end up in a “grey zone,” which requires special attention from medical writers, researchers, and sponsors.
The ICMJE criteria serve as the global benchmark for biomedical authorship, providing a framework that researchers rely on to maintain publishing integrity. The problem, however, is that while clear, these criteria still leave room for uncertainty.
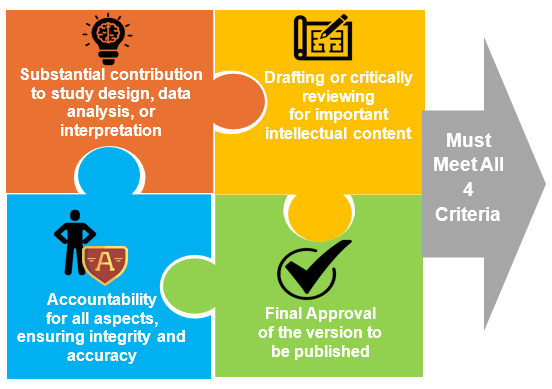
Gold Standard: ICMJE 4 Pillars Test
The ICMJE continues to be the guiding light. For someone to be considered an author, they have to satisfy all four requirements1.
Acknowledgments: Recognizing Support1
An acknowledgment is used to recognize the contribution of those who did not contribute to the study enough to be authors. Typically, these people include:

While these are necessary roles, but they do not involve direct intellectual ownership of the manuscript. However, distinguishing between “support” and “substantial contribution” is where the grey zone emerges.
It should be noted that professional medical writers, unless they satisfy all ICMJE guidelines, are normally acknowledged as opposed to being listed as authors. Transparency is crucial, and as recommended by Good Publication Practice (GPP 2022), it is essential to disclose the type of writing assistance received and its funding.
The Grey Zone: Where Boundaries Blur
Despite having well-defined boundaries, there are various situations which complicate the black-and-white classification of contributors’ status:
1. Medical Writers as Authors vs. Acknowledged Contributors
Medical writers frequently make a huge contribution in drafting manuscripts, shaping narratives, and ensuring compliance with journal standards. However, drafting alone does not qualify for authorship unless accompanied by intellectual contributions to study design or data interpretation2.
This creates a grey zone:
- If a writer contributes intellectually (e.g., interpreting results), authorship may be justified.
- If their role is limited to structuring or editing, acknowledgment is appropriate.
The problem is how to objectively measure an “intellectual contribution”.
2. Data Contributors Without Writing Involvement
Researchers involved in data collection or patient recruitment often expect authorship. However, ICMJE explicitly states that data acquisition alone is insufficient. This leads to tension, especially in multicenter studies where contributors may feel under-recognized3.
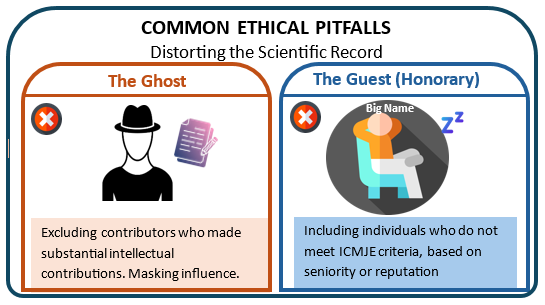
3. Senior Leadership and “Honorary Authorship”
Including department heads or senior investigators who have minimal involvement remains a persistent ethical concern. Such “guest” or “honorary” authorship violates ICMJE principles and undermines accountability4.
4. Ghost writing and Under-Acknowledgment
At the opposite end is ghost-writing, where contributors (often medical writers) are neither listed as authors nor acknowledged. This lack of transparency raises serious ethical and regulatory concerns, particularly in industry-sponsored research5.
Why This Grey Zone Matters

Authorship is not just recognition—it is a declaration of responsibility for the scientific integrity of the work2.
Emerging Solutions to Reduce Ambiguity
To address this grey zone, several approaches are gaining traction:

Conclusion
The line dividing authorship and acknowledgment in the realm of medical writings is not always clearly defined. Although the guidelines provided by the ICMJE present an excellent framework for consideration, practical circumstances can sometimes blur the line in ways that call for critical thinking. The increasing need for transparency and accountability in the modern era of science cannot be overlooked.
Ultimately, the goal is not just to assign credit, but to uphold the integrity of scientific communication.
Note: The image featured in this article is generated using NotebookLM for illustration purposes.
References
- (2026). Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals. http://www.icmje.org/icmje-recommendations.pdf.
- International Committee of Medical Journal Editors. Defining the role of authors and contributors [Internet]. International Committee of Medical Journal Editors; 2025 [cited 2026 Apr 23]. Available from: https://icmje.acponline.org/recommendations/browse/roles-and-responsibilities/defining-the-role-of-authors-and-contributors.html.
- Savage KD. Information for contributors. Appl Biosaf. 2019 Dec 1;24(4):231–238. doi:10.1177/1535676019873741.
- Meursinge Reynders, R.A., ter Riet, G., Di Girolamo, N. et al. Honorary authorship is highly prevalent in health sciences: systematic review and meta-analysis of surveys. Sci Rep 14, 4385 (2024). https://doi.org/10.1038/s41598-024-54909-w.
- Jung-Choi K. Research misconduct using a ghostwriter in a paper-writing company. Ann Occup Environ Med. 2023 Nov 7;35:e45. doi:10.35371/aoem.2023.35.e45.
- Partin K, Hosseini M. Using the contributor role taxonomy (CRediT) as a tool in resolving authorship disputes at the NIH. Account Res. 2025 Dec 7:2596063. doi:10.1080/08989621.2025.2596063.
- DeTora LM, Toroser D, Sykes A, Vanderlinden C, Plunkett FJ, Lane T, et al. Good Publication Practice (GPP) guidelines for company-sponsored biomedical research: 2022 update. Ann Intern Med. 2022 Sep;175(9):1298–1304. doi:10.7326/M22-1460.
Author:

Manasvi Joshi, Msc in Biotechnology
Senior Scientific Writer 1, Enago Life Sciences
Connect with Manasvi on Linkedin


