Linguistic Validation Service that Leaves No Room for Errors in the Launch of Your Clinical Trial.
Linguistic validation is vital in multinational clinical trials. Enago Life Science's clinical expertise ensures seamless adaptation of sensitive content across languages and cultures. We help meet stringent standards for Clinical Outcome Assessment (COA) validation, following FDA and ISPOR procedures. Our commitment ensures conceptual, cultural, and constructive equivalence in all targeted languages.

PhD
in Medicinal Chemistry, The Central University of Venezuela
24+ Years of Experience


MS
Microbiology, Southern Illinois University, USA
20+ Years of Experience


PhD
in Medical Sciences, Nagoya University, Japan
13+ Years of Experience


Doctor
Dental Medicine, Public Health, Universidade de Lins, Brazil
12+ Years of Experience


Doctor
of Pharmacy (Pharm.D.), Pharmacy and Biochemistry
10+ Years of Experience

Some of the Documents That Require Linguistic Validation
- Patient-Reported Outcome (PRO) Instruments
- Clinical Trial Documents
- Healthcare Questionnaires and Forms
- Regulatory Documents
- Pharmaceutical Labels and Packaging
- Educational Materials
- Medical Device
Instructions - Training Materials
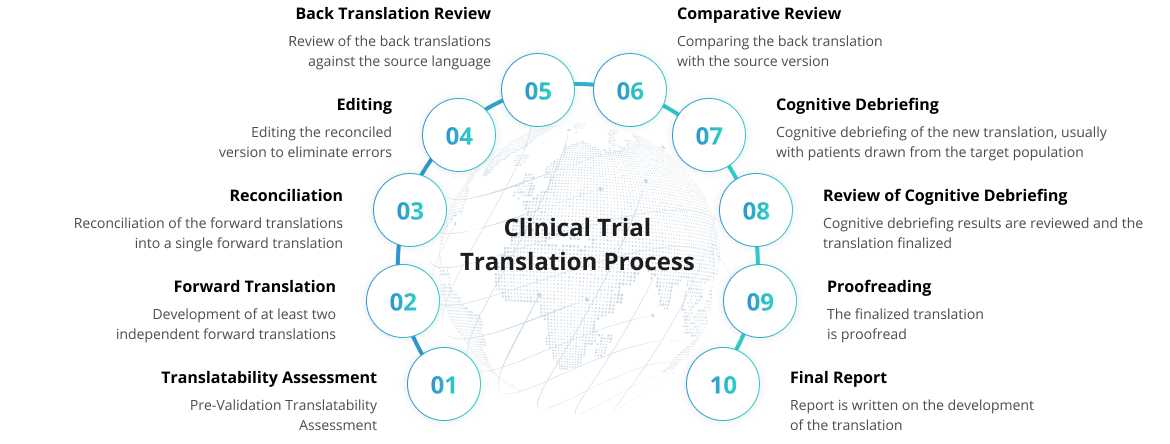
Certified Processes That Ensure Superior Quality
of
Translated Outcomes
Recognizing the significance of quality, we are committed to providing services that
consistently meet the highest standards of excellence.
200+
Languages
125+
Countries
3k+
Language Experts
200k+
Clients Worldwide
Why Select Enago Life Science for Your COA Translations?
We've designed our localization experience with certified processes and efficient technology, ensuring a seamless delivery of consistent quality that enhances client satisfaction.

Specialized Subject-
Area Matching

Quality-
Driven Technology

24x7 Dedicated
Localization Manager

ROI-Driven
Localization Solutions

In-House Expertise

ISO-Certified Processes
Ensure best quality translation in any language through linguistic validation done by medical experts.
GET IN TOUCHWhat do our clients say?

Through their excellent project management system, Enago Life Sciences surpassed all my expectations and passed fairly. Given the demands of this project, it was difficult to meet my requirements but Enago Life Sciences successfully delivered my work following the timelines and high-quality standards. It is a miracle company.


Enago Life Sciences completed 400,000+ words of translation in 40 business days, all delivered on time; at the same time, the translation quality is also fully achieved to the level required by Alvogen (Lotus). We are happy and satisfied to collaborate with Enago Life Sciences. Thank you Enago Life Sciences team.


Enago Life Sciences’ translation and localization services have been highly convenient for us and I am grateful to the entire team for being so generous and sincere towards our requirements. At this point, we can almost blindly trust them with everything. They barely needed our supervision and the result delivered was exactly what we were looking for. Honestly, what else could we ask for! Thank you, Enago Life Sciences. You’re simply the best!


Working with Enago Life Sciences was the best decision, as we did not have to worry about any issue. We want to thank team Enago Life Sciences for giving us their continuous support. They went out of their way to ensure that we are satisfied with the project which impressed us as well as members at the Fukushima Medical University. Thank you all once again for doing a fantastic job on this challenging project and handling it with the same passion as we felt when compiling the book.


I have been using Enago Life Sciences services regularly for some time now and their quality of work is really good. The style and accuracy with which they translated my various documents was exactly what I expected it to be. I am really impressed with their team who worked closely with me to translate our vision into reality.

Our Memberships