Modern precision medicine integrates genetic risk, pharmacogenetics, epigenetics, microbiome data, mobile health data, and electronic health records to tailor treatments at the individual level. While this integration has significantly advanced clinical research and care, it has also introduced the challenge of simplifying complex data.
As highlighted in a study, precision medicine relies on combining heterogeneous data sources to generate clinically relevant insights, but these datasets are often difficult to interpret, even for trained experts. This shift has redefined a core challenge in life sciences. It is no longer just about generating high-quality data, but about translating that data into clear, actionable insights for a diverse group of stakeholders including clinicians, researchers, regulators, payers, and patients.
Why Effective Communication Matters More Than Ever
1. Stakeholders Interpret the Same Data Differently
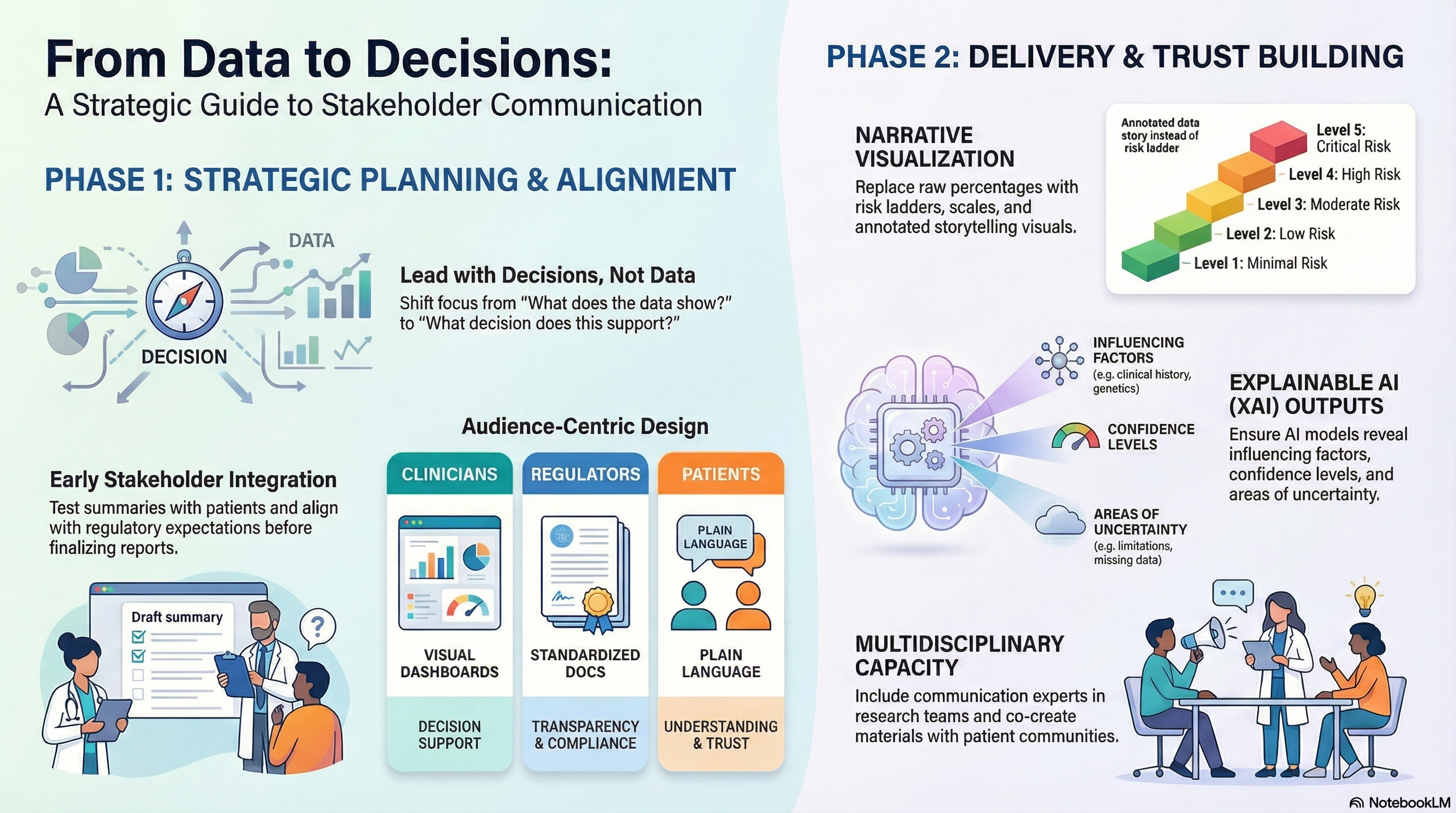
Precision medicine requires coordination across stakeholders with varying expertise and expectations. This is further supported by a qualitative study which found that effective implementation depends on aligning expectations across researchers, clinicians, policymakers, and patients.
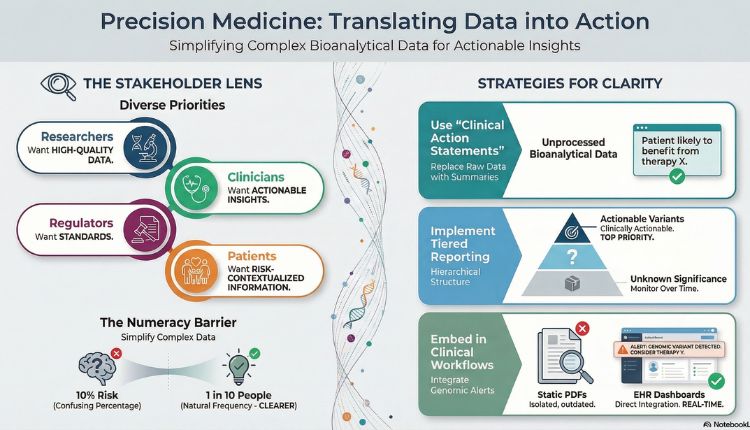
However, each stakeholder interprets the same dataset through different priorities:
- Researchers focus on methodological transparency and reproducibility
- Clinicians prioritize actionable insights and clinical relevance
- Regulators look for standardized, auditable data
- Patients need simple, understandable, risk-contextualized information
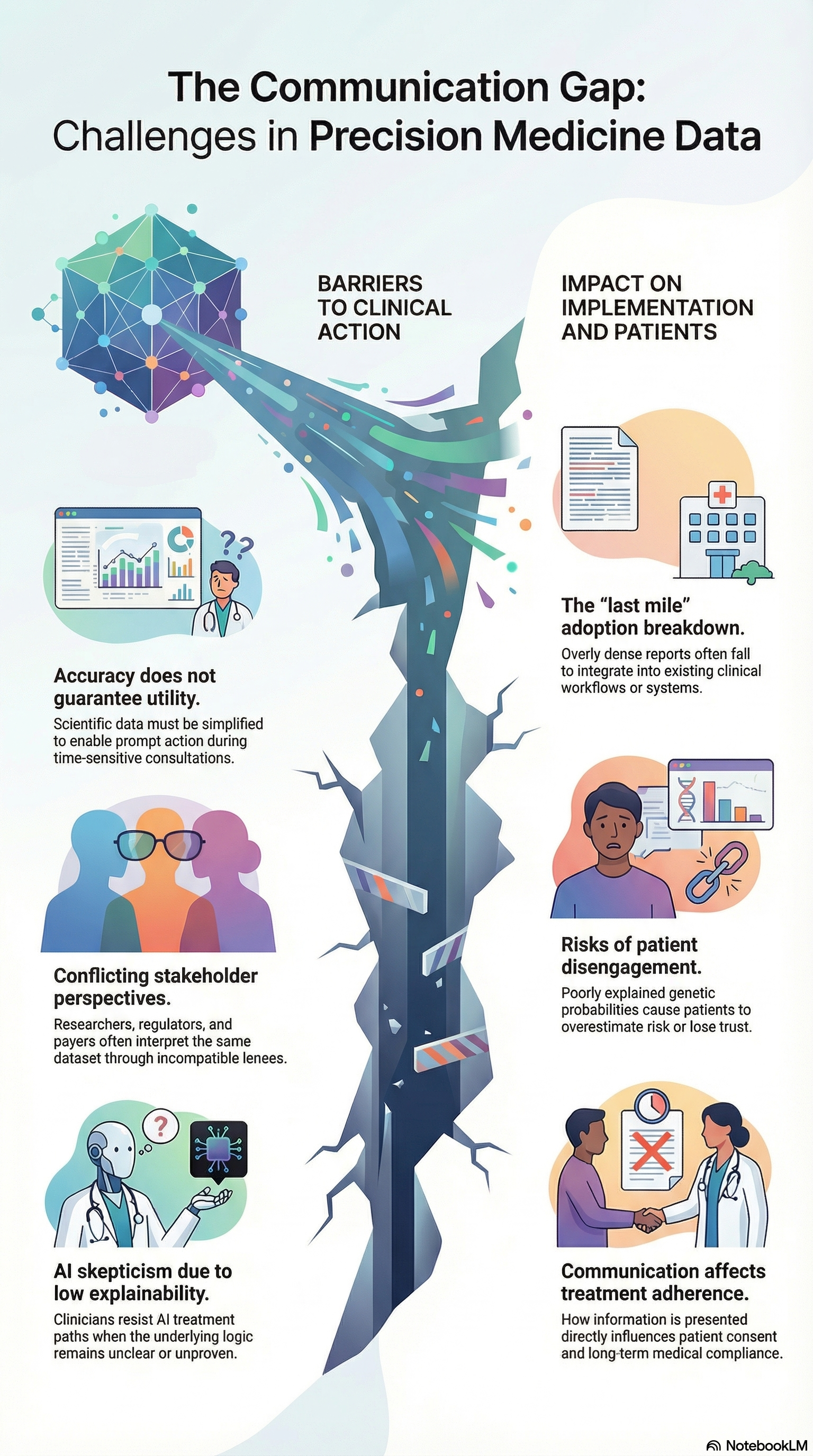
When communication does not account for these differences, interpretations diverge. This slows down decision-making and limits real-world impact.
2. Limited Health Numeracy Among Patients
Communicating risk remains a persistent challenge. Research shows that health numeracy (the ability to understand quantitative health information) is often limited among patients. This places the burden on communicators to simplify information without distorting meaning. For example, probabilistic risk (e.g., “20% increased risk”) is often misinterpreted, and concepts like randomization or genomic variants require contextual explanation.
3. Growing Pace of Data Complexity Over Communication Practices
Biomedical data ecosystems are becoming increasingly fragmented, with persistent challenges around data silos, heterogeneity, and integration. At the same time, precision medicine is increasingly driven by multi-omics datasets, machine learning and AI-based models, and probabilistic and predictive outputs.
Note: The above image is generated using NotebookLM for illustration and understanding purposes.
While these advances improve analytical power, they make communication significantly more difficult, particularly across interdisciplinary teams. Addressing these challenges does not require simplifying the science, it requires rethinking how insights are communicated. In practice, several strategies are emerging that help bridge the gap between complexity and clarity.
Strategies for Effective Communication
1. Convert Analytical Outputs into “Clinical Action Statements”
One of the most effective shifts in clinical genomics and precision oncology reporting is the move from raw data to structured action statements. Instead of presenting variant classifications, hazard ratios, and model outputs, leading institutions now include clear, decision-oriented summaries, such as:
“This patient is likely to benefit from EGFR-targeted therapy based on detected mutation X.”
This approach aligns with recommendations from the American College of Medical Genetics (ACMG)and Genomics, which emphasize interpretation and clinical relevance alongside raw findings.
How to implement:
- Add a “Clinical Interpretation” section to reports
- Use standardized language for actionability (e.g., “recommended,” “consider,” “insufficient evidence”)
- Pair every key result with a “what this means” statement
2. Use Tiered Reporting Models
Many healthcare systems have moved toward tiered or layered reporting, especially in oncology and pharmacogenomics. For example, cancer genomic reports increasingly follow a structure like:
- Tier 1: Clinically actionable variants
- Tier 2: Potential clinical significance
- Tier 3: Variants of unknown significance
This model is recommended by the Association for Molecular Pathology (AMP) and has been shown to improve interpretability for clinicians.
How to implement:
- Categorize findings based on clinical relevance
- Prioritize actionable insights at the top
- Clearly separate exploratory or uncertain findings
3. Integrate Outputs Directly into Clinical Workflows
A major reason precision medicine insights go unused is that they exist outside clinician workflows. Embedding genomic and clinical decision-support outputs into electronic health records (EHRs) can significantly improve utilization and adherence. For example, the eMERGE Network demonstrated that integrating genomic alerts into EHR systems improved clinical uptake of pharmacogenomic recommendations.
How to implement:
- Integrate reports into EHR dashboards instead of PDFs
- Use alerts for high-impact findings
- Ensure outputs are accessible at the point of care
4. Apply Evidence-Based Risk Communication Formats for Patients
Patient misunderstanding of risk is well documented, but validated formats exist that improve comprehension. For example,
- Absolute risk is better understood than relative risk
- Natural frequencies (e.g., “2 out of 10”) are clearer than percentages
- Visual aids significantly improve understanding
How to implement:
- Replace “20% increased risk” with “2 out of 10 people may…”
- Use icon arrays or risk ladders
- Add a one-line interpretation: “This means your risk is slightly higher than average”
5. Use Standardized, Machine-Readable Reporting Frameworks
To improve reproducibility and regulatory alignment, structured formats like BioCompute Objects (BCOs) are increasingly used to communicate complex bioinformatics workflows. This can standardize how genomic analyses are documented and shared.
How to implement:
- Use structured templates for bioanalytical workflows
- Ensure traceability of data sources and methods
- Enable machine-readable outputs for regulatory submissions
6. Build Explainability Layers into AI Outputs (Not Just Performance Metrics)
In clinical AI, high accuracy alone is not sufficient; it also requires transparency to ensure credibility and reproducibility.
How to implement:
- Include feature importance summaries in outputs
- Provide confidence scores and uncertainty ranges
- Add “why this prediction was made” sections
7. Test Communication Materials with Real Users Before Deployment
One of the most overlooked yet high-impact strategies is usability testing.
Healthcare organizations increasingly conduct:
- Clinician usability testing for reports
- Patient comprehension testing for summaries
Studies show that iterative testing significantly improves understanding and reduces misinterpretation.
How to implement:
- Run small pilot tests with clinicians before scaling
- Conduct patient interviews or surveys
- Track comprehension and decision accuracy
8. Use Real-World Case-Based Communication
During COVID-19 and in oncology decision making, scenario-based communication proved highly effective.
Instead of abstract data, reports framed insights as:
- “If untreated vs. treated” scenarios
- “Best-case vs. worst-case outcomes”
This approach improves both clinician decision-making and patient understanding.
How to implement:
- Include scenario comparisons in reports
- Use “what happens next” pathways
- Provide context, not just data
In practice, effective communication in precision medicine is not about simplifying the science, it is about structuring it in ways that align with how different stakeholders make decisions.
![Communicating precision medical data across stakeholders]() Note: The above image is generated using NotebookLM for illustration and understanding purposes.
Note: The above image is generated using NotebookLM for illustration and understanding purposes.
This avoids the common scenario where perfectly good data has to be reworked (sometimes multiple times) because it wasn’t designed with the end user in mind.
Precision medicine promises transformative impact, but only if its insights are understood and trusted by those who use them. The future of bioanalysis is not just about better data, but about better communication of that data.
Communication can no longer be treated as a downstream activity; something that happens after analysis is complete. It must be built into the process itself, shaping how data is structured, interpreted, and delivered from the outset.
The organizations that will lead in this space are not necessarily those with access to the largest datasets, but those that can translate complexity into clarity without losing scientific rigor. Whether it is enabling clinicians to make faster decisions, helping regulators evaluate evidence more efficiently, or empowering patients to engage meaningfully with their care, effective communication sits at the center of impact.
In many ways, the future of precision medicine will be defined not just by how precisely we can analyze data, but by how clearly we can explain it.
Author:

Anagha Nair
Editorial Assistant, Enago Academy
Medical Writer, Enago Life Sciences
Connect with Anagha on LinkedIn


 Note: The above image is generated using NotebookLM for illustration and understanding purposes.
Note: The above image is generated using NotebookLM for illustration and understanding purposes.