Introduction:
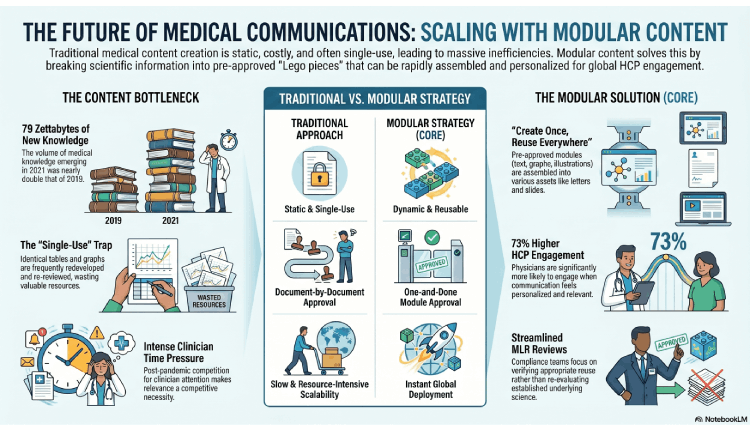
The time pressure on clinicians has intensified since the COVID-19 pandemic, and the competition for their limited attention has also grown sharply 1. At the same time, the volume of medical knowledge being published is expanding at a staggering rate. An Elsevier survey of 1778 physicians found that 79 zettabytes of new medical knowledge emerged in 2021—almost twice the 2019 volume1. In this environment, the Medical Affairs teams can no longer sustain creating unique content for every channel, region, and audience. The industry’s answer is modular content—pre-approved, reusable blocks of scientific information assembled, adapted, and deployed at scale. Done well, modular content drives efficiency, global scalability, and true omnichannel HCP engagement.
What Is Modular Content and Why Does It Matter?
Modular content uses pre-approved blocks of content called “modules” to build medical assets like medical information letters (MILs), scientific exchange materials, and medical education materials 1. Rather than recreating multiple versions of the same deliverable, core messages, graphs, and illustrations are centrally crafted, reviewed, and stored in a library —ready for local teams to adapt 1.
The inefficiency of the traditional approach is well-documented. Medical Affairs content is static, costly, and often single use. Even identical elements, for example, a table from a published manuscript reused in a slide deck, are frequently redeveloped and re-reviewed each time 1. This duplication breeds inconsistency, delays delivery, and consumes resources that could be better spent on evidence generation and stakeholder engagement. The modular approach resolves this by enabling a “Create Once, Reuse Everywhere” (CORE) philosophy. Biopharma Medical Affairs teams pioneered modular content to handle multiple product launches and to boost content output without increasing resources. By turning content into reusable “Lego pieces” and standardizing assembly, biopharma has improved both the speed and consistency of its content operations 1.
The Global Scalability Imperative
 Scaling content in the pharmaceutical world isn’t about “doing more”—it’s about doing things smarter. Here is a breakdown of how the big players are ditching the old, slow ways of communication for a modular approach:
Scaling content in the pharmaceutical world isn’t about “doing more”—it’s about doing things smarter. Here is a breakdown of how the big players are ditching the old, slow ways of communication for a modular approach:
- The “One and Done” Approval: Instead of getting every single document approved individually, companies are moving toward modular content. You approve a piece of information once, and then you can legally and safely reuse it across apps, chatbots, and websites2.
- Quality Over Quantity: The real secret to global growth isn’t drowning your audience in more “stuff.” It’s building a smarter infrastructure where content is designed to be broken down, swapped around, and updated instantly across dozens of markets 2.
- The Physician Preference: The data is clear—73% of physicians are more likely to engage when the communication feels personal. In an industry where time is the most precious commodity, being relevant isn’t just a “nice to have”; it’s a competitive necessity 1.
- A Massive Global Shift: This isn’t just a theory. Global leaders have already rolled this out across 20+ markets, retraining hundreds of people to stop thinking in “documents” and start thinking in “modules.”
The Architecture Behind Scalable Modular Content
Achieving global scalability with modular content requires deliberate architecture across three dimensions: content strategy, governance, and technology.
1. Data-Led Content Strategy

Data is the heartbeat of a great content strategy. This means understanding HCP needs and usage patterns, mapping out required deliverables, and aligning content plans with customer journey touchpoints across channels. The goal isn’t to create a static document. Instead, you build a living plan that evolves as fast as the data does. When new insights are generated, the content plan must be updated to reflect them1. It’s about being agile, staying relevant, and ensuring that every piece of information you share serves a genuine purpose.
2. Anticipating Content Governance:

The biggest hurdle for modular content is governance that includes meeting regional regulatory requirements, copyright considerations, and the Medical Legal Regulatory (MLR) review process 1. Compliance, legal, and regulatory partners should be brought to the table early during strategic and content planning stages. The goal is to review materials through an omnichannel lens from the outset, avoiding repetitive re-review of the same content for different purposes and audiences. When compliance teams understand the full intended use of a piece of content, they can streamline their review approach accordingly 1. When we respect those regional differences up front, we create a system that is both fast and incredibly safe 1.
3. Technology: DAM as the Backbone

A well-organized digital asset management (DAM) system is essential for power content automation. A central, searchable repository allows users to filter and find global content for local reuse or channel-specific deployment. Metadata, taxonomy, and standardized fields are core components of effective DAM: they define and sort assets, and without them, even the best content library becomes difficult to navigate. The lesson from organizations that have built these systems is to strategize before configuring1.
Overcoming the Barriers: A Practical Starting Point
The most common barriers include
- organizational complexity
- regulatory caution
- technology investment thresholds
Steps to overcome barriers:
- Pragmatic Start: You don’t need a complex IT infrastructure to begin; start by identifying high-value, frequently reused scientific content and building a pre-approved repository around it. Get those components reviewed and approved 3.
- Efficiency Gains: Review cycles become faster because compliance teams focus on verifying appropriate reuse rather than re-evaluating the underlying science 3.
- Strategic Writing: Use standardized terminology, editorial style, and short, self-contained statements instead of narrative flows to ensure pieces can stand alone.
- Flexible Visuals: Avoid embedding text in illustrations to make them easier to translate and repurpose across different channels 1.
The Strategic Opportunity
The pharmaceutical industry is shifting toward a unified HCP experience, with Medical Affairs leading the transition from reactive, ad hoc content creation to proactive modular content operations. This modular infrastructure acts like a “navigable river,” allowing scientific data to flow consistently across all channels—from medical information to commercial engagement 1. Beyond efficiency, this foundation enables true personalization: delivering the right scientific insights to the right HCP at the perfect moment. That is what meaningful Medical Affairs engagement looks like in the omnichannel era.
References
- Falcone B, Riggins J, D’Ambrosia M. Fran Paradiso Hardy, Vice President, Global Medical Communications, Astellas Pharma Inc.
- Modular Content: Driving Change in Pharma Content Operations | The Stem. Accessed April 23, 2026. https://www.thestem.com/casestudy/modular-content-driving-change-in-pharma-content-operations/
- Mellums H. How to Overcome the Challenge of Modular Content in Medical Communications. MedComms Experts. January 22, 2026. Accessed April 23, 2026. https://www.medcomms-experts.com/modular-content-alternative-medical-affairs/
Author:

Sweaksha Langoo (MSc. Molecular Biology and Biochemistry)
Scientific Writer – Enago Life Sciences
Connect with Sweaksha on LinkedIn